|
This is strictly true for all elements in the s and p blocks. 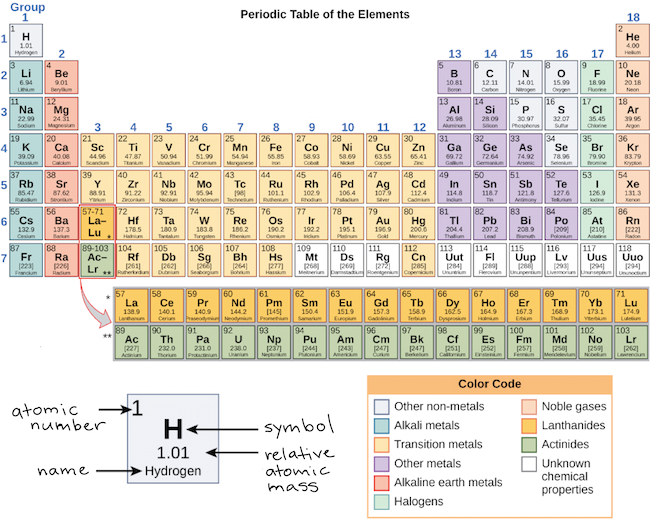 
Elements in each column have the same valence shell electron configurations, and the elements have some similar chemical properties. The same concept applies to the other columns of the periodic table. The organization of electrons in atoms explains not only the shape of the periodic table, but also the fact that elements in the same column of the periodic table have similar chemistry. From the elements position on the periodic table, predict the valence shell electron configuration for each atom. Tins electron configuration is Kr5 s 2 4 d 10 5 p 2. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry- and they do. Sn is located in the second column of the p block, so we expect that its electron configuration would end in p 2. They all have a similar electron configuration in their valence shells: a single s electron. Lewis, suggested a simple way to keep track of the number of valence electrons: draw dots around the symbol of an element to. The second column on the right has the different corresponding electron configurations for the specified element in the rows underneath. However, there are also other patterns in chemical properties on the periodic table. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. The first column on the left has various elements in the rows underneath. Elements in each column have the same valence shell electron configurations, and the elements have some similar chemical properties. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Table with two columns and 6 rows. Remember, The half filled and fully filled orbitals are more stable and the elements try to get this configuration. If Zinc leaves the electrons from its 4s orbital, then It'll have 4s0 3d10 configuration. Thus, the periodic table becomes a tool for remembering the charges on many ions. For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. If Zinc leaves the electrons from its 3d orbital, then It'll have 4s2 3d8 configuration. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. Introduction Valence Electrons and the Periodic Table Tyler DeWitt 1.4M subscribers Subscribe Subscribed 22K 1.9M views 10 years ago To see all my Chemistry videos, check out. If we look at just the valence shell's electron configuration, we find that in each column, the valence shell's electron configuration is the same. Valence is the number of connections an atom tends to form. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. Scientists soon observed patterns in the valence of the different elements. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. S block is blue, d block is green, p block is red and f block is orange. Empty periodic table, separated into 4 different colored blocks. The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs.\): Blocks on the Periodic Table. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. As we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner orbitals. By locating the element on the periodic table, the table is sectioned off into s,p,d, and f blocks. Elements are either metals, nonmetals, or semimetals. How do you find the valence electron configuration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
